Over the past weeks there have been several exciting updates in COVID-19 clinical trials. The first are some results released from the ACTT-2 trial, which is evaluating combined Remdesivir with Barcitinib (a JAK inhibitor). This trial, in collaboration with Eli Lily, reported beneficial outcomes of the combined therapy in reducing COVID-19mortality by 35% (by Day 29, p=0.04) , reducing average time to recovery from 8 to 7 days, and improving overall clinical outcomes (defined as time to discharge, p=0.04). Given positive results of combined therapy in the ACTT-2 trial, there is hope on the …
mRNA-1273 Induced Robust Immune Responses in a Small Cohort of Older Adults
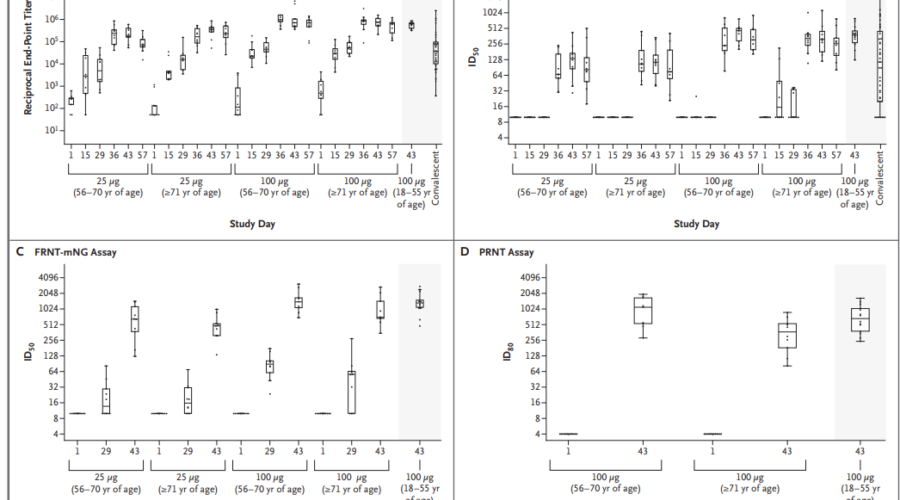
Summary: Results of a phase 1 trial have revealed that the Covid-19 vaccine developed by the US-based biotechnology company Moderna elicited a strong immune response in older adults with no serious adverse effects. Last week, Moderna released the results of the expansion of their phase 1 trial of mRNA-1273, one of the more prominent COVID vaccine candidates in trials, which included the preliminary safety and immunogenicity data for healthy participants who were 56 years of age or older (Table 1). The trial followed similar dosing and antibody response analysis protocols from their previous …
Pfizer’s new antiviral, the fourth phase III trial for a SARS-CoV-2 vaccine begins, and NIH expansion of convalescent plasma trials for COVID-19
There have been many advances in clinical trials over the past two weeks. The first large announcement is Pfizer’s antiviral, PF-07304814, is entering phase I trials (NCT04535167). This antiviral was originally developed in the SARS-CoV-1 outbreak, but was ended prematurely in trials due to a lack of demand. However, Pfizer has reported in a pre-print that the antiviral has efficacy across multiple coronaviruses including SARS-CoV-2, although this pre-print has yet to withstand peer review (Boras B, et al. 2020 ). With Remdesivir being the only antiviral to demonstrate efficacy in SARS-CoV-2 …