As we move ever closer to that ultimate triumph of science over nature – a successful vaccine for COVID-19 – it is increasingly important that we, and especially those in the business of vaccine development, harbor an understanding of how the biology of SARS-CoV-2 interacts with our own, and how we may leverage that interaction to create the most effective vaccine possible. In an article published last month in Nature Reviews Immunology, Jeyanathan et al. outline some of the most important considerations for vaccine development, including the nature of the host immune response, existing …
mRNA-1273 Induced Robust Immune Responses in a Small Cohort of Older Adults
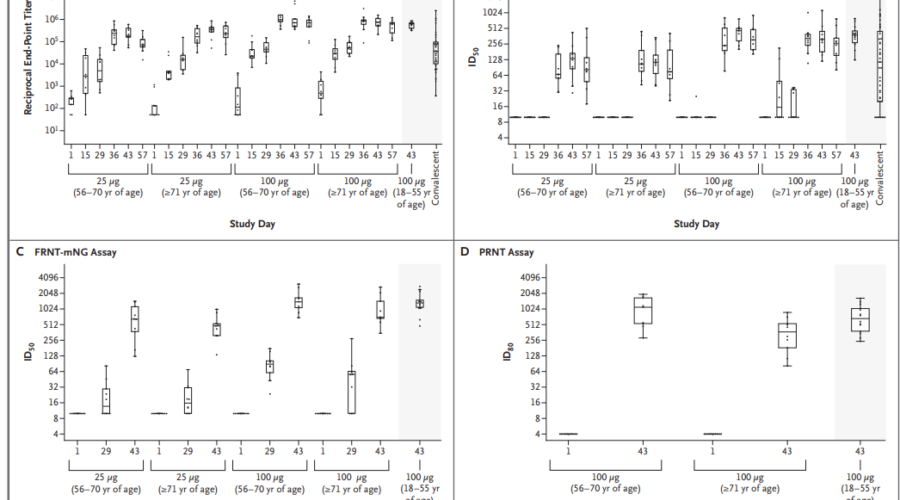
Summary: Results of a phase 1 trial have revealed that the Covid-19 vaccine developed by the US-based biotechnology company Moderna elicited a strong immune response in older adults with no serious adverse effects. Last week, Moderna released the results of the expansion of their phase 1 trial of mRNA-1273, one of the more prominent COVID vaccine candidates in trials, which included the preliminary safety and immunogenicity data for healthy participants who were 56 years of age or older (Table 1). The trial followed similar dosing and antibody response analysis protocols from their previous …
US Government Announces SARS-CoV-2 Vaccine Distribution Strategy
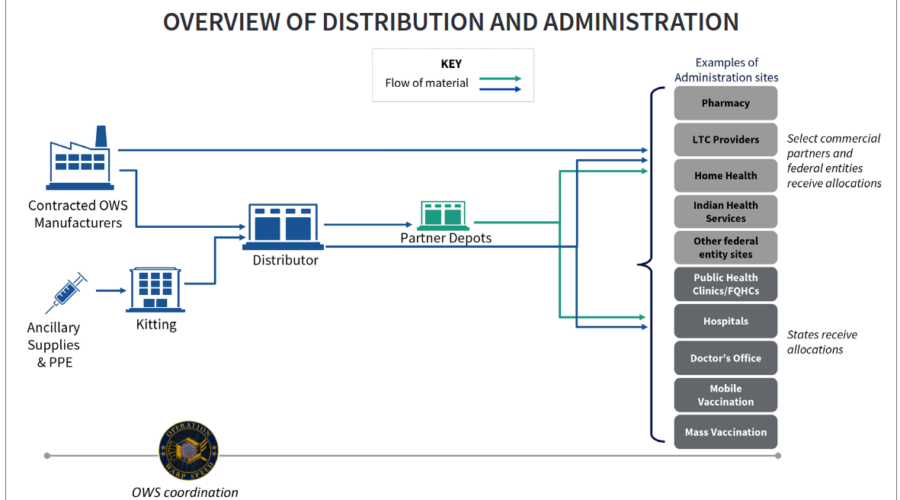
Last week, the Trump administration released guidelines regarding the distribution strategy for a COVID-19 vaccine through the U.S. Department of Health and Human Services (HHS) and the Department of Defense (DoD). These guidelines, found in this link, detail the American government’s plan to safely and effectively distribute a vaccine ahead of time as many of the developers in the market are still working to get their candidate approved (with the first yet to be approved in the U.S. as the time of this report). In addition, the guidelines assure that the administration aims to provide the …