Large-scale protein level comparison of Deltaproteobacteria reveals cohesive metabolic groups
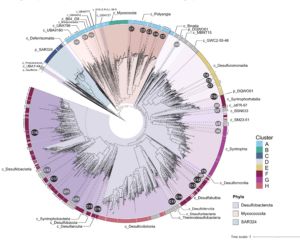
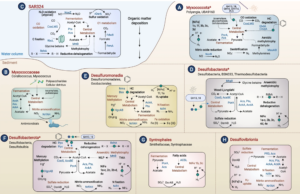
Deltaproteobacteria, now proposed to be the phyla Desulfobacterota, Myxococcota, and SAR324, are ubiquitous in marine environments and play essential roles in global carbon, sulfur, and nutrient cycling. Despite their importance, our understanding of these bacteria is biased towards cultured organisms. Here we address this gap by compiling a genomic catalog of 1,792 genomes, including 402 newly reconstructed and characterized metagenome-assembled genomes (MAGs) from coastal and deep-sea sediments. Phylogenomic analyses reveal that many of these novel MAGs are uncultured representatives of Myxococcota and Desulfobacterota that are understudied. To better characterize Deltaproteobacteria diversity, metabolism, and ecology, we clustered ~1,500 genomes based on the presence/absence patterns of their protein families. Protein content analysis coupled with large-scale metabolic reconstructions separates eight genomic clusters of Deltaproteobacteria with unique metabolic profiles. While these eight clusters largely correspond to phylogeny, there are exceptions where more distantly related organisms appear to have similar ecological roles and closely related organisms have distinct protein content. Our analyses have identified previously unrecognized roles in the cycling of methylamines and denitrification among uncultured Deltaproteobacteria. This new view of Deltaproteobacteria diversity expands our understanding of these dominant bacteria and highlights metabolic abilities across diverse taxa.