New paper on the metabolic diversity of Thorarchaeota
Comparative genomic inference suggests mixotrophic lifestyle for Thorarchaeota
New paper on the metabolic diversity of ultrasmall archaea
On August 25, 2017 my laboratory took a direct hit by a category 4 hurricane, Harvey.
Here is some of the press that covered the impacts of the storm on my lab, institute, lab members, and family.
Nature – How labs are coping with Hurricane Harvey’s devastating floods
Science – A lab flees from Harvey: We were ‘just so damn lucky’

New paper published, resolving the metabolisms of the dead zone.
New paper detailing hydrocarbon and nutrient cycling in deep sea hydrothermal sediments
Congrats Nina and Kiley!
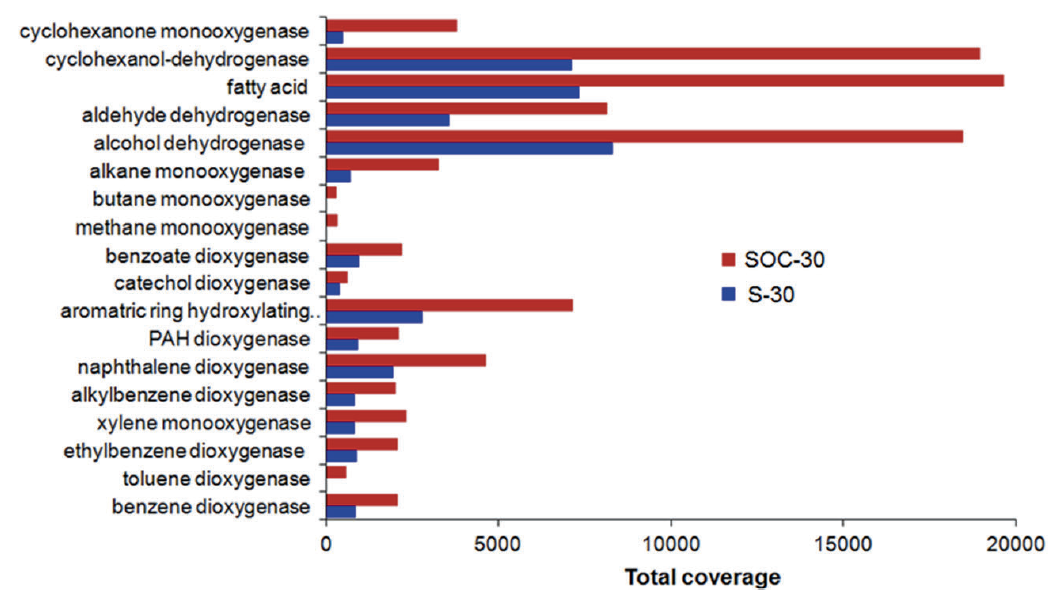
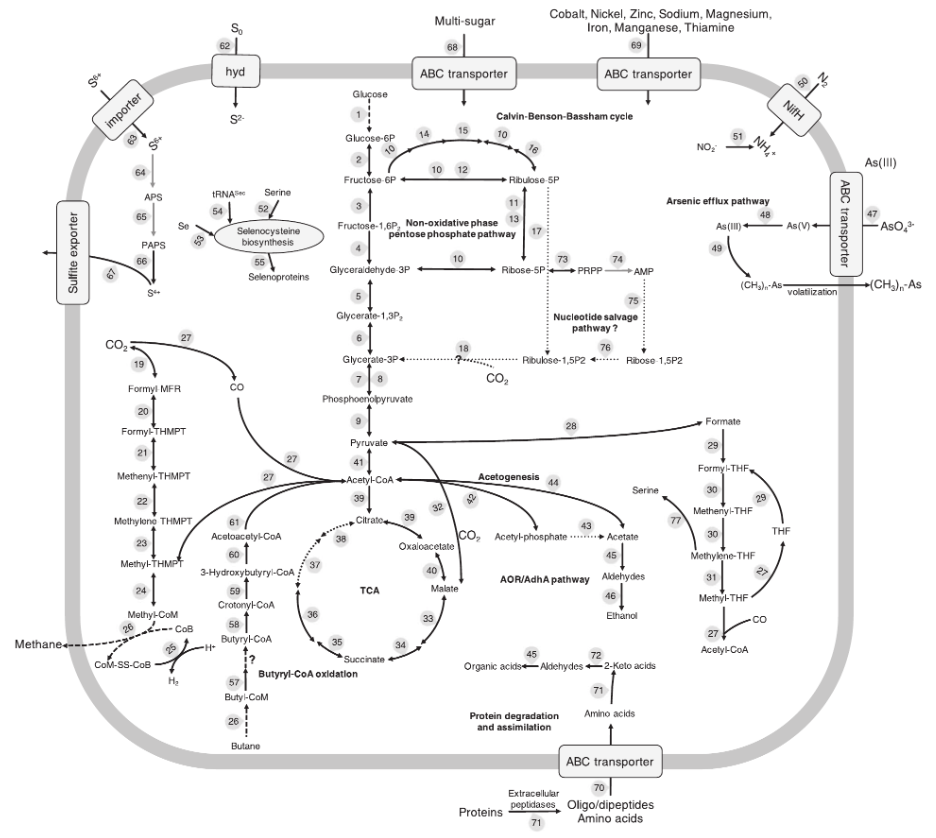
This paper details the genetic diversity of these sediments and describes genomes belonging to a uncultured archaeal group (GoM-Arc1) that contain novel pathways for hydrocarbon cycling, related to ANME (anaerobic methane oxidizers).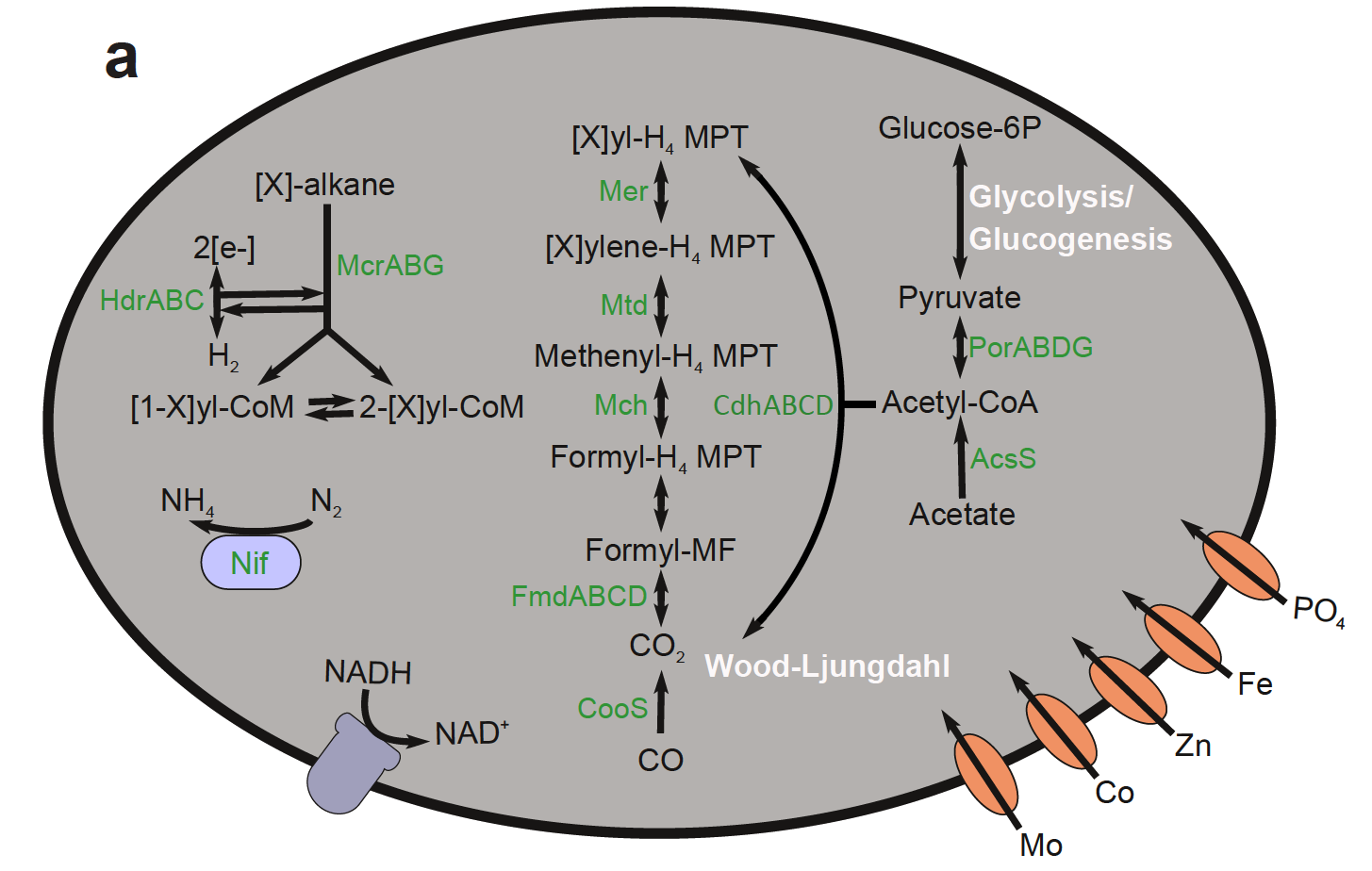
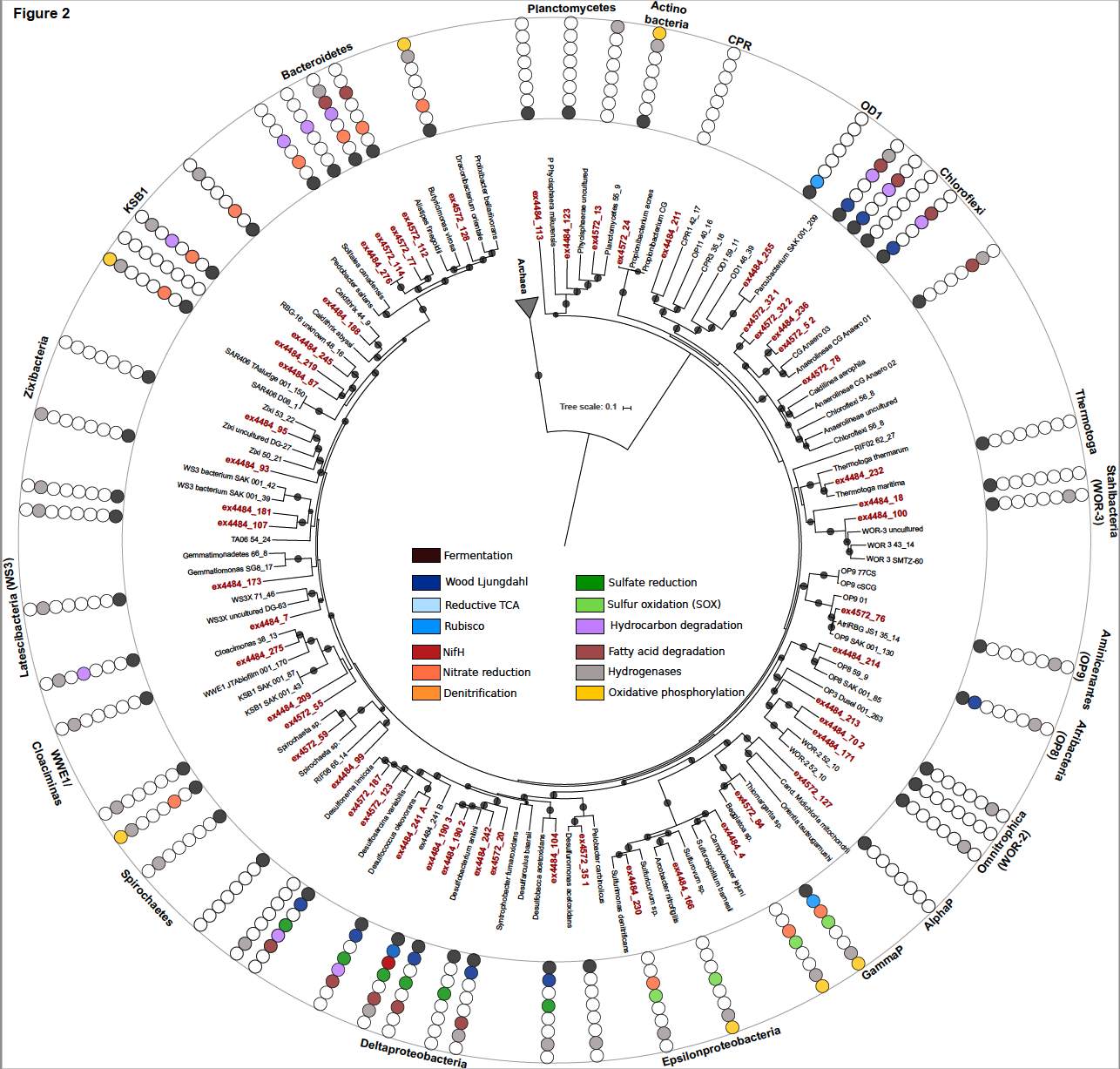
Introducing a new class in the archaea, Theionarchaea, and several other uncultured lineages
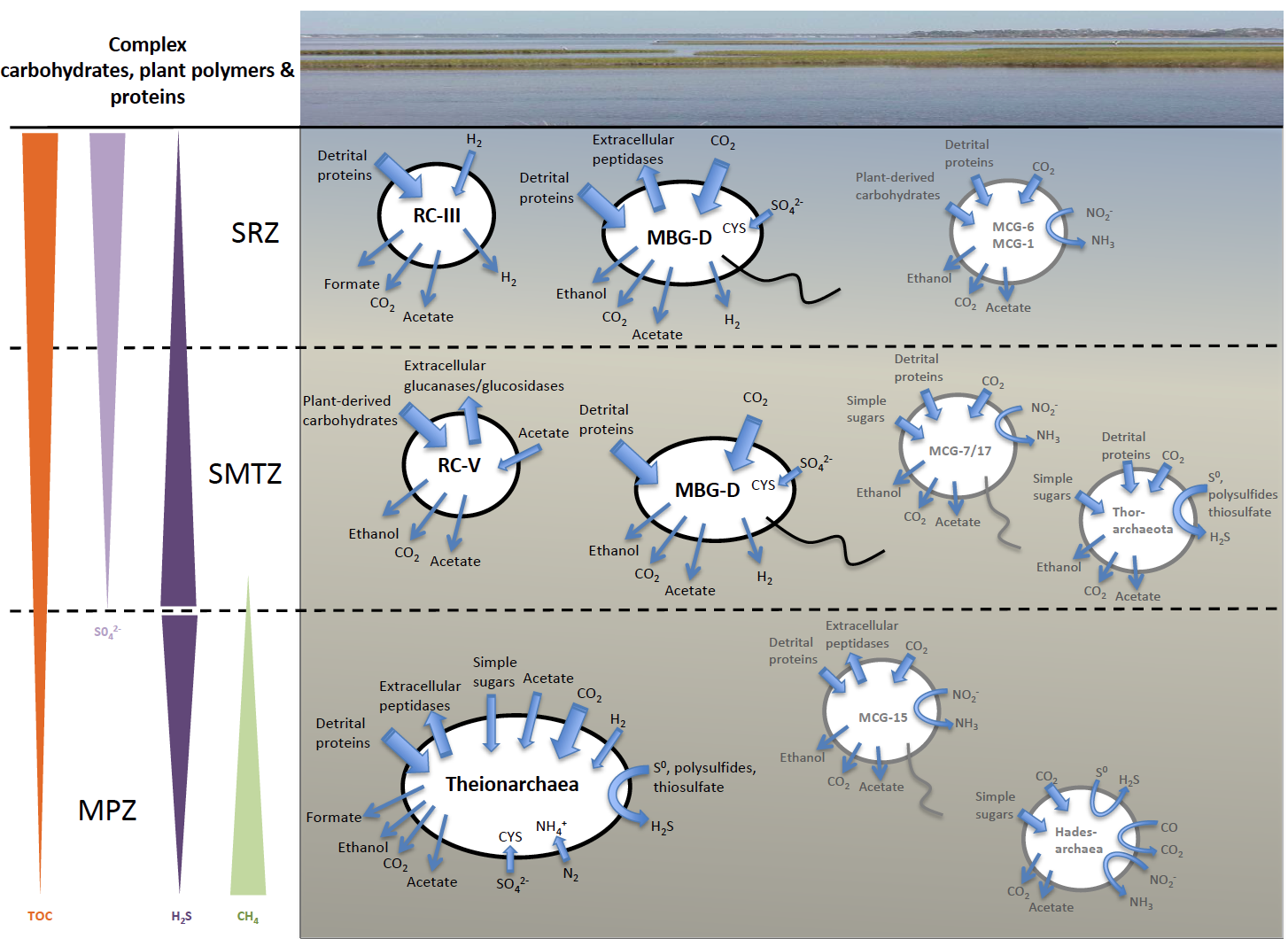
In addition to Theionarchaea, this new paper that appears in ISME Journal also details a variety of archaeal genomes there were obtained from the White Oak River Estuary in North Carolina. This digram summarizes the ecological roles we have inferred from these genomes. This is important because NONE of these lineages have been grown in a laboratory, so having their genomes has significantly advanced our understanding of what they do in nature.
Introducing Asgard! A new superphylum of archaea that are related to eukaryotes.
Asgard archaea illuminate the origin of eukaryotic cellular complexity

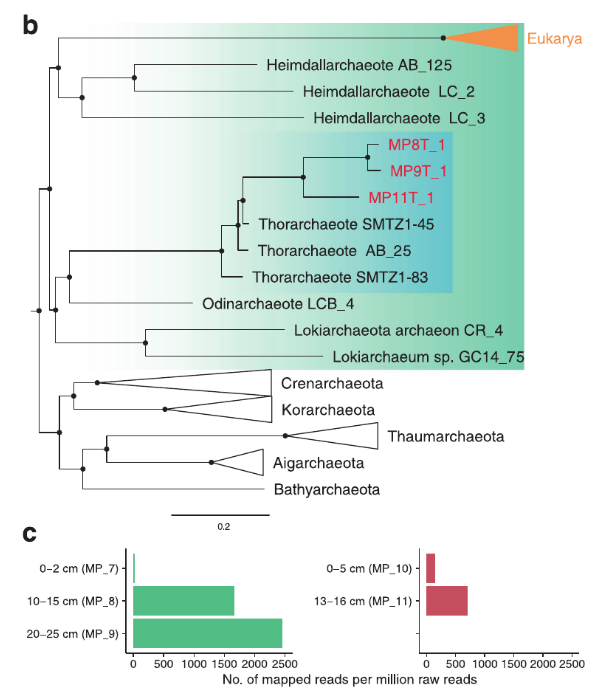
 This week our new paper describing the discovery of four archaea phyla that are related to eukaryotes was published in Nature. These phyla belong to the same branch of life and have been named after different Norse gods, Thor, Odin, Heimdall, and Loki. This is a collaboration with Thijs Ettema’s lab in Sweden. Last year we published the discovery of Thorarchaeota in ISME.
This week our new paper describing the discovery of four archaea phyla that are related to eukaryotes was published in Nature. These phyla belong to the same branch of life and have been named after different Norse gods, Thor, Odin, Heimdall, and Loki. This is a collaboration with Thijs Ettema’s lab in Sweden. Last year we published the discovery of Thorarchaeota in ISME.
This paper adds two additional phyla, Odinarchaeota and Heimdallarchaeota. The focus of this paper is to further resolve the phylogenetic position of eukaryotes in this new superphylum. It also examines the presence of several new ESPs or eukaryotic signature proteins. These proteins were mostly thought to exist in eukaryotes, but these genomes contain a variety of them!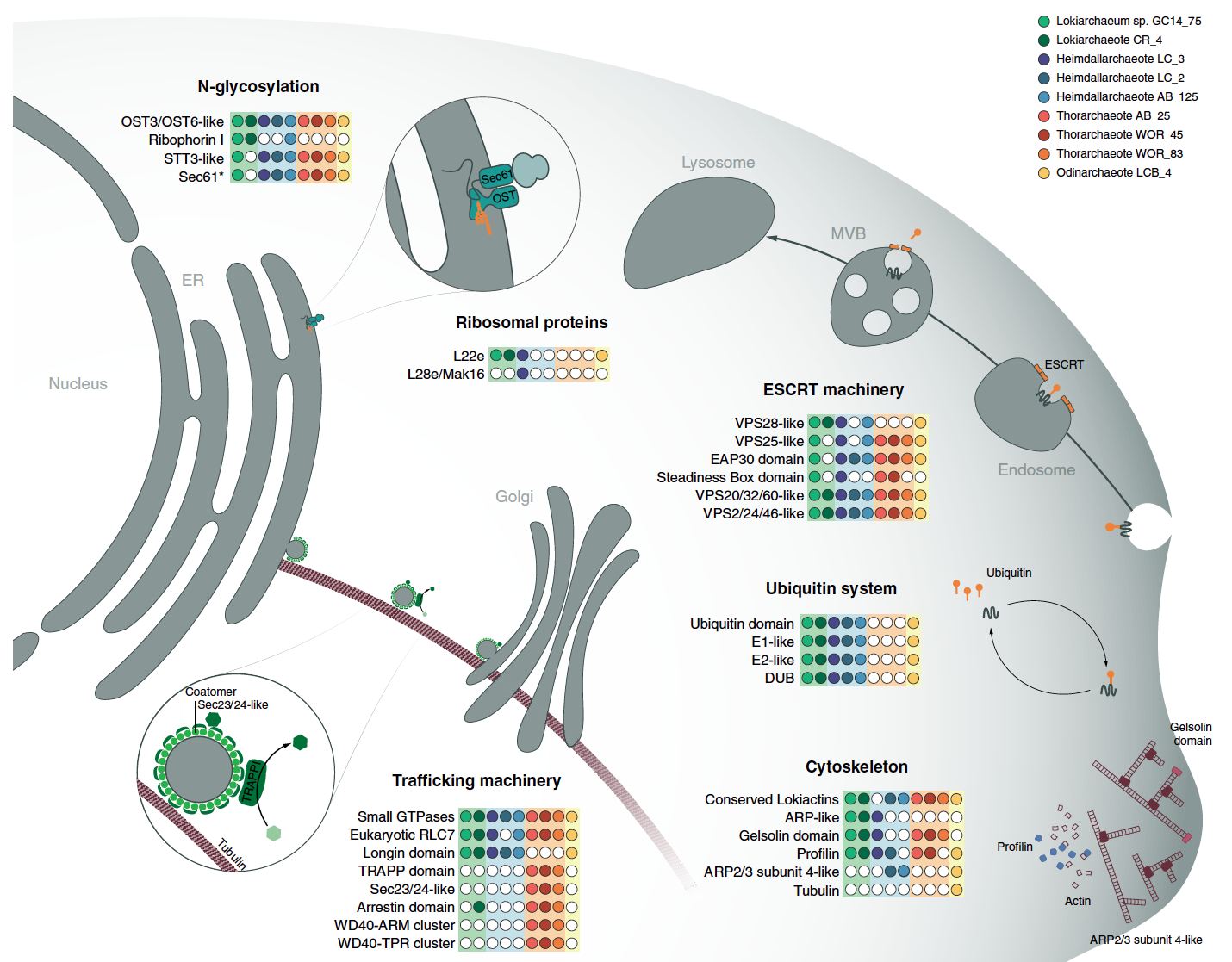
Press releases to accompany this study:
A nice News and Views article about our recent publication in Nature Microbiology.
Marine Microbiology: a community cleanup
by Rachel Mackelprang & Olivia U. Mason
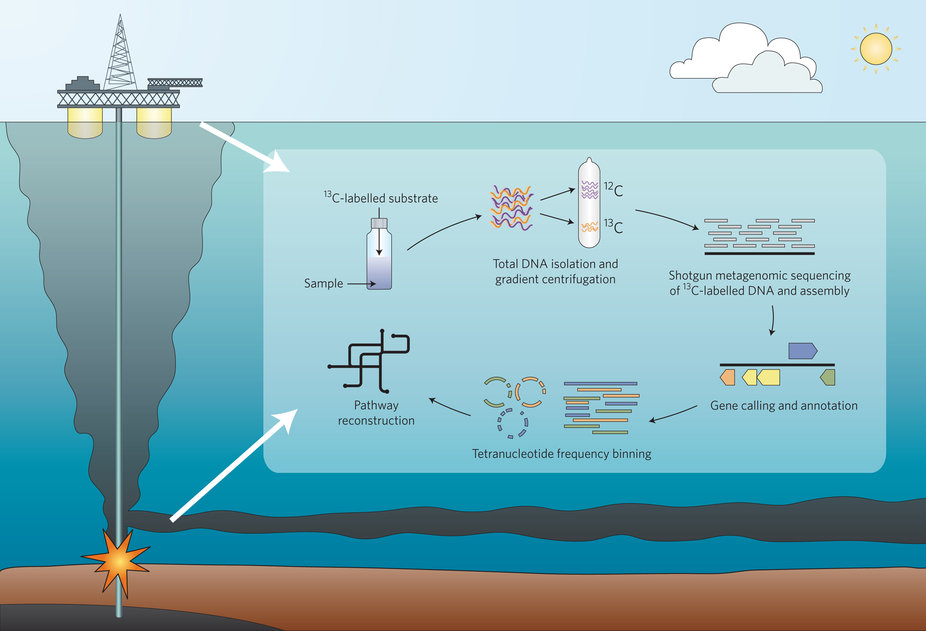 DNA-SIP metagenomic experimental strategy for the identification and characterization of hydrocarbon-degrading microorgansims from DWH oil spill deep-plume and surface-slick samples.
DNA-SIP metagenomic experimental strategy for the identification and characterization of hydrocarbon-degrading microorgansims from DWH oil spill deep-plume and surface-slick samples.